For a long time, incandescent bulbs were the main source of artificial light in our homes and streets. By heating a filament until it glowed, they produced the warm light that defined everyday lighting for more than a century. Today, however, most everyday lighting relies on LEDs, which are far more energy-efficient and longer-lasting. This change marks one of the most important advances in everyday lighting.
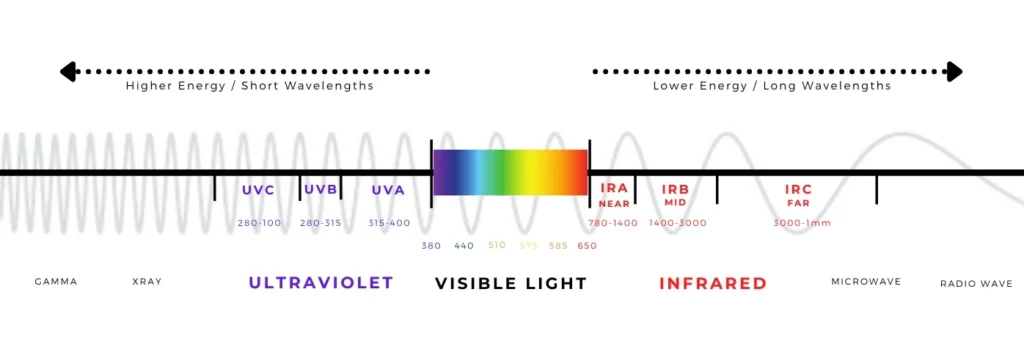
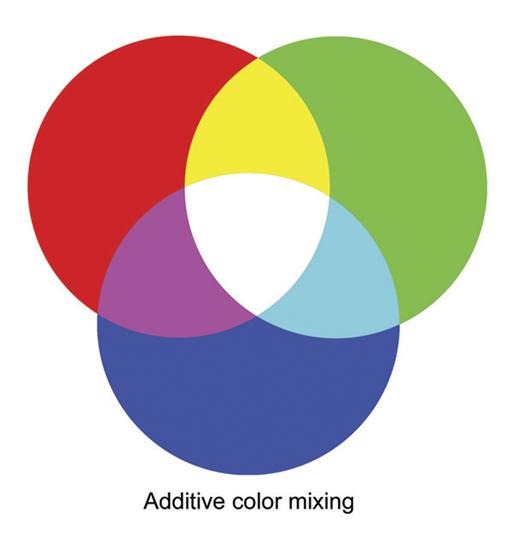
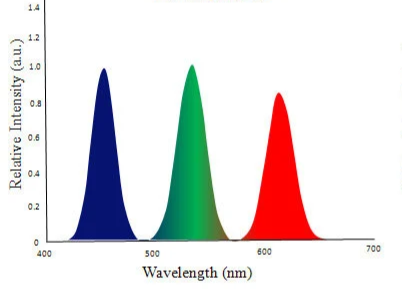
First, we need to understand how we perceive colour. Light is part of the electromagnetic spectrum (Figure 1), but our eyes can detect only a small portion of it: visible light. Objects appear coloured because they absorb some wavelengths of light and reflect others into our eyes. In additive colour mixing, combining the three primary colours of light (red, green, and blue) produces white light (Figure 2). Some displays and specialised LED systems work in exactly this way, by carefully mixing red, green, and blue emitters (Figure 3). However, this is usually not how white LEDs for everyday lighting are made, since that approach is more complex and often less efficient for general illumination.
Instead, photonics offers a much more elegant mechanism: fluorescence. In simple terms, fluorescence occurs when a material absorbs light of one colour, usually one with higher energy, which means it is closer to the blue end of the spectrum, and then re-emits part of that energy as light of another colour, usually with lower energy, closer to the red end. A familiar example can be seen in fluorescent paints or minerals that glow under ultraviolet light (Figure 4). The key idea is that the material transforms the incoming light into new wavelengths, changing the colour we ultimately perceive.

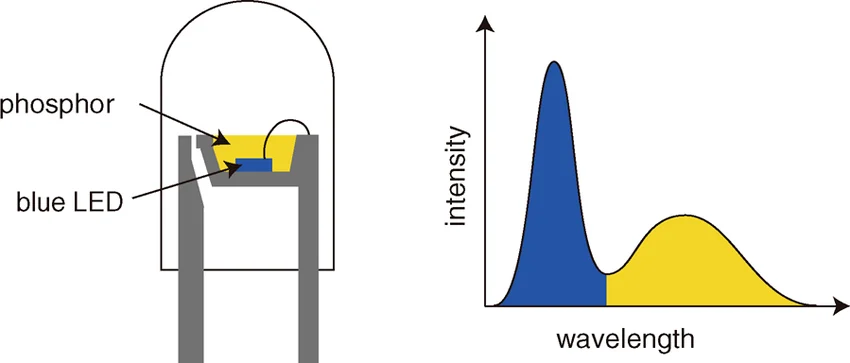
This is exactly the principle behind most white LED bulbs. They are not truly “white” light sources by themselves. In most cases, the heart of the device is actually a blue LED. This blue light then passes through a fluorescent coating, typically made of phosphor materials, which absorbs part of the blue emission and re-emits it at longer wavelengths, especially in the yellow region (Figure 5). The combination of the remaining blue light and the converted yellowish light is perceived by our eyes as white. So, in a sense, many white LEDs are indeed based on blue light, but transformed through fluorescence into something much richer.
In the end, white LEDs are a beautiful example of how modern photonics turns a simple physical effect into a technology that shapes everyday life. What appears to be ordinary white light is actually the result of a clever interaction between a blue semiconductor and fluorescent materials. Rather than a trivial engineering trick, it is a reminder that even the technologies we use every day can be built on fascinating physics.
By: Borja García García
